Lines Spectra and Excited Electron States
By A Mystery Man Writer
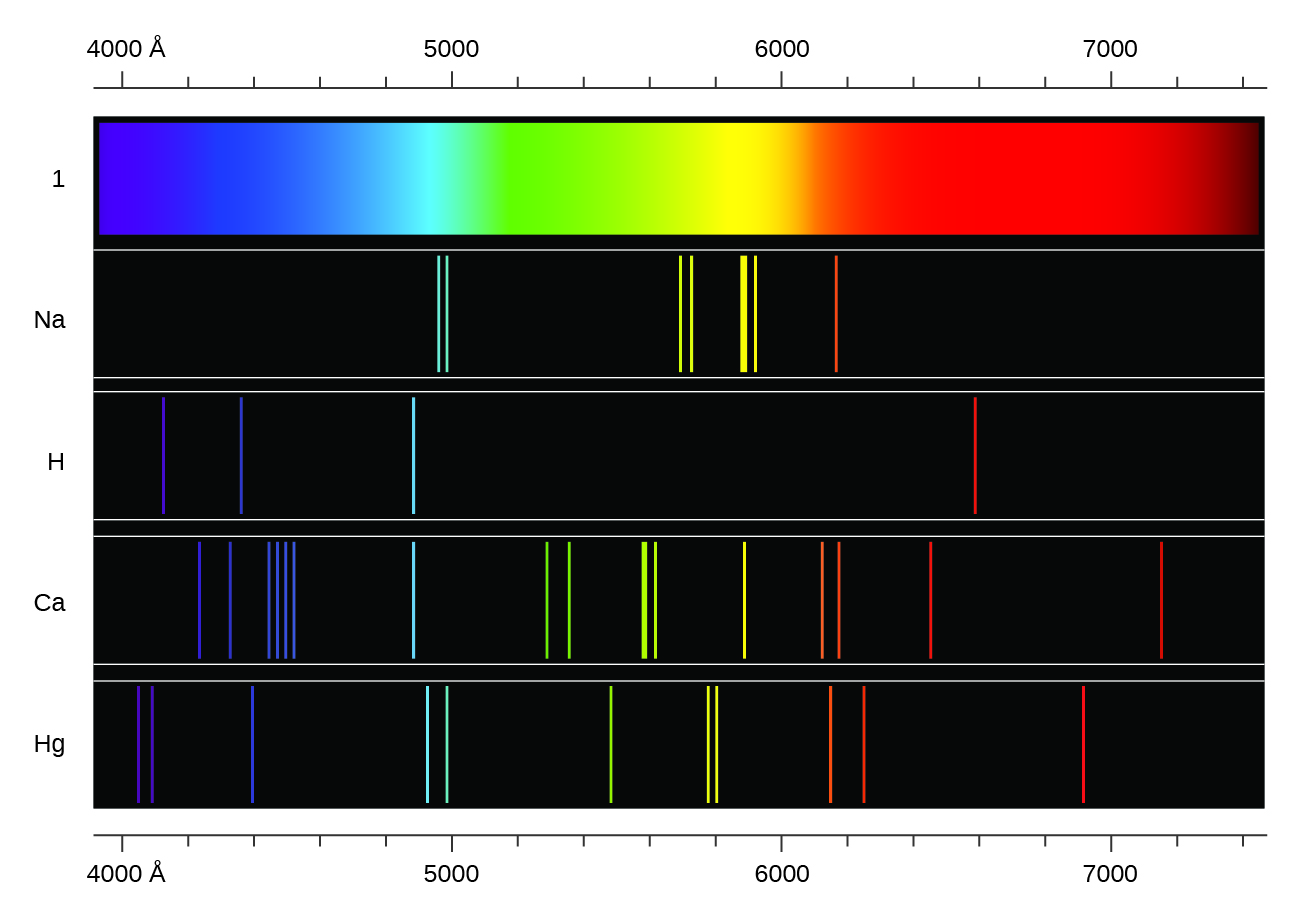

Machine learning lets researchers see beyond the spectrum
Spectroscopy: Interaction of light and matter (article)
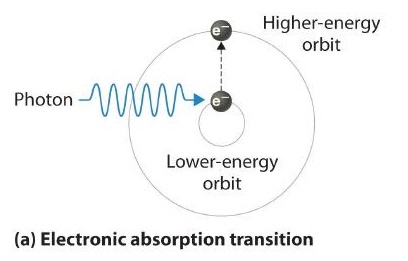
6.3: Line Spectra and the Bohr Model - Chemistry LibreTexts
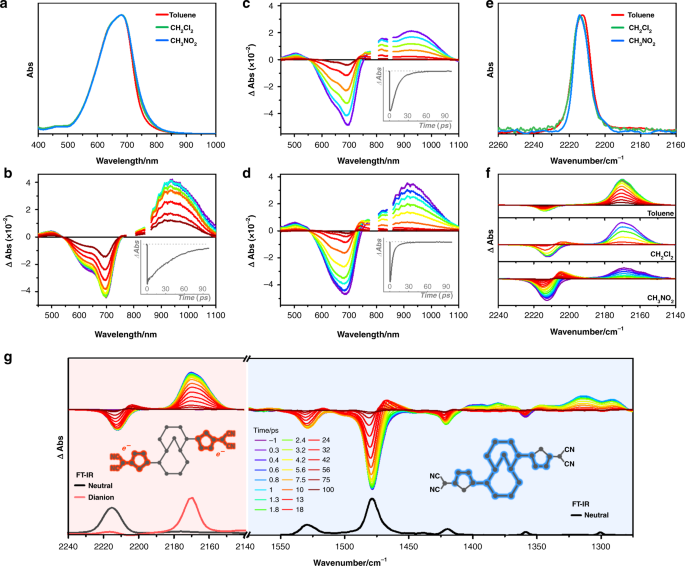
Two-electron transfer stabilized by excited-state aromatization
A sample contains only hydrogen atoms and in all atom electron are present at 4th excited state. How many minimum number of atoms are required if total 7 lines are observed in

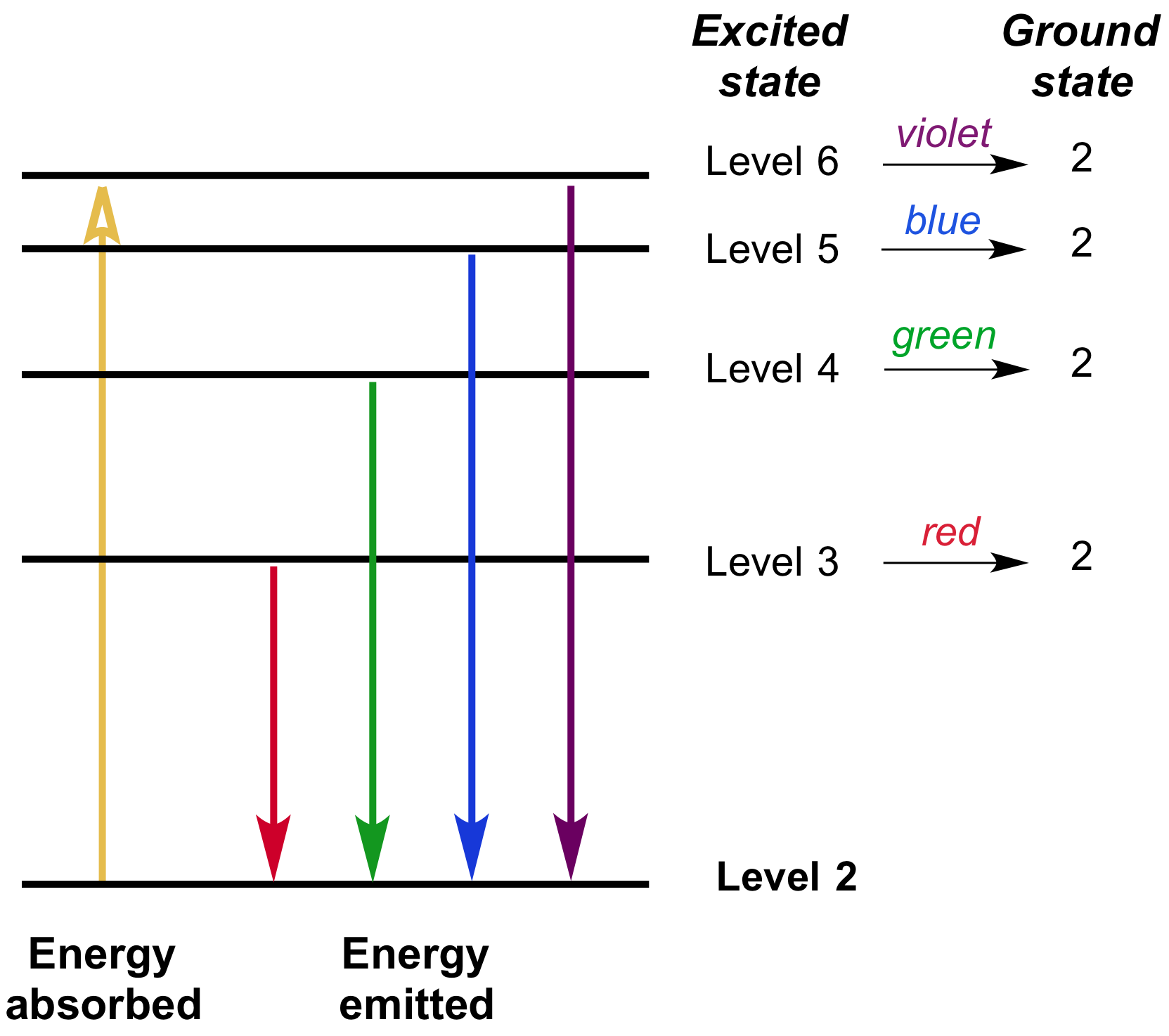
Formation of Spectral Lines
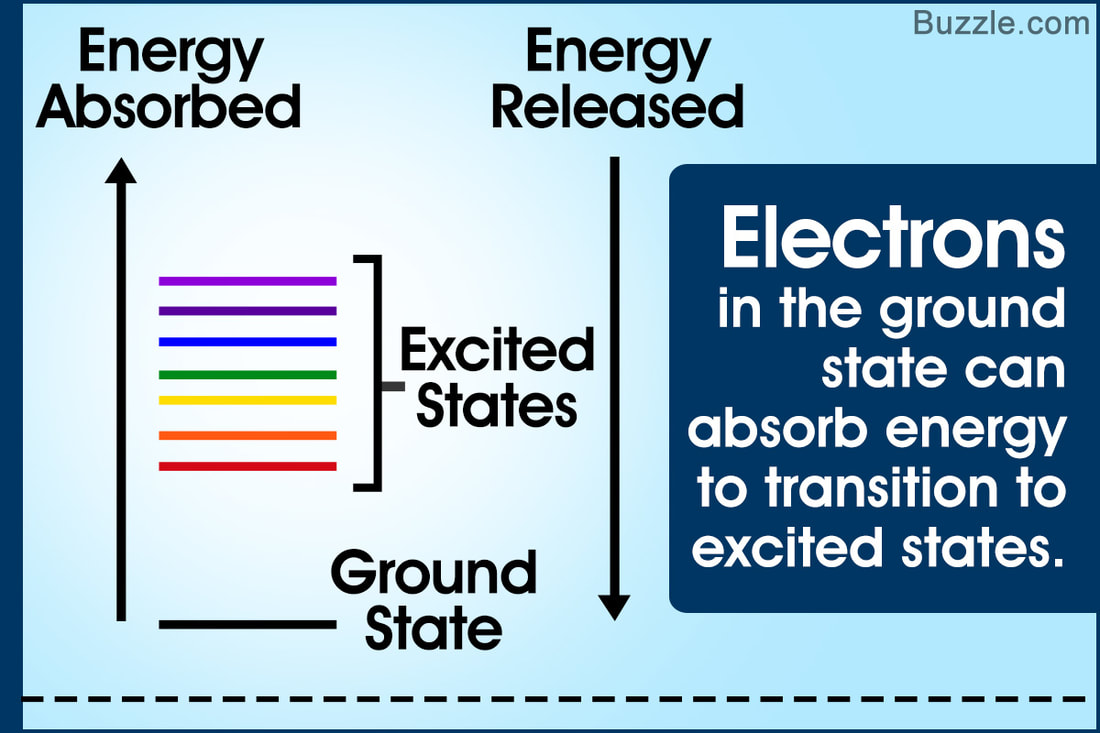
CHEM 4.1.U2

Lesson Explainer: Emission and Absorption Spectra
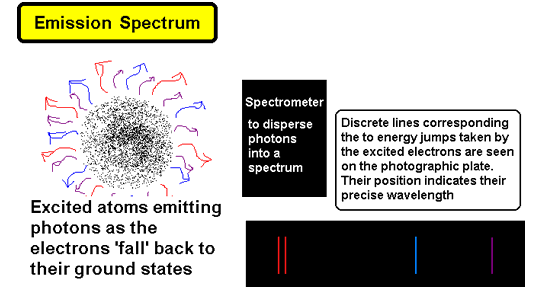
Atomic Spectra - 'fingerprints' for elements

The figure below represents part of the emission spectrum for a one-electron ion in the gas phase. All the lines result from electronic transitions from excited states to the n = 3